As04 atropine antidote12/15/2023 Use only the liquid medication in children under 13 years old do not use tablets. If no response within 48 hours, this medicine is unlikely to be effective. Reduce dose as soon as initial control of symptoms has been achieved. 
These doses are approximate adjust downward according to nutritional status and degree of dehydration. Maintenance dose: Once control is achieved, reduce dose to individual requirements, to as little as one quarter of the initial daily dosage Initial dose: 0.3 to 0.4 mg/kg of diphenoxylate, orally, in 4 divided doses If no improvement is seen within 10 days, symptoms are unlikely to respond to further administration. Clinical improvement is usually seen within 48 hours. Maximum dose: 20 mg diphenoxylate per day Maintenance dose: Once control is achieved, reduce dose to individual requirements, to as little as 2 tablets or 10 mL once a day Initial dose: 2 tablets or 10 mL (5 mg of diphenoxylate), orally, 4 times a day You may report side effects to FDA at 1-80.Ītropine and diphenoxylate dosing information 
Call your doctor for medical advice about side effects. This is not a complete list of side effects and others may occur. Nausea, vomiting, upset stomach, loss of appetite or Rapid breathing, weak or shallow breathing ĭehydration symptoms-feeling very thirsty or hot, being unable to urinate, heavy sweating, or hot and dry skin.Ĭommon side effects of atropine and diphenoxylate may include:Ĭonfusion, feelings of extreme happiness Severe pain in your upper stomach spreading to your back įever, flushing (warmth, redness, or tingly feeling) Severe constipation, stomach pain or bloating Stop using this medicine and call your doctor at once if you have: This medicine may cause serious side effects. Some side effects may occur up to 30 hours after you take atropine and diphenoxylate. Antidote : Atropine Sulfate Gastrointestinal Complication Paralytic ileus. All rights reserved.Get emergency medical help if you have signs of an allergic reaction: hives difficult breathing swelling of your face, lips, tongue, or throat. In conclusion, ATR/OBI has been proven to be efficacious against exposure to VX and there were no PK interactions between ATR and OBI when administered as a co-formulation.Ītropine sulfate Guinea pig Nerve agents Obidoxime chloride Pharmacokinetics.Ĭopyright © 2020 The Authors. Moreover, AChE activity after VX exposure in both blood and brain tissue was significantly higher in ATR/OBI-treated animals compared to vehicle-treated control. 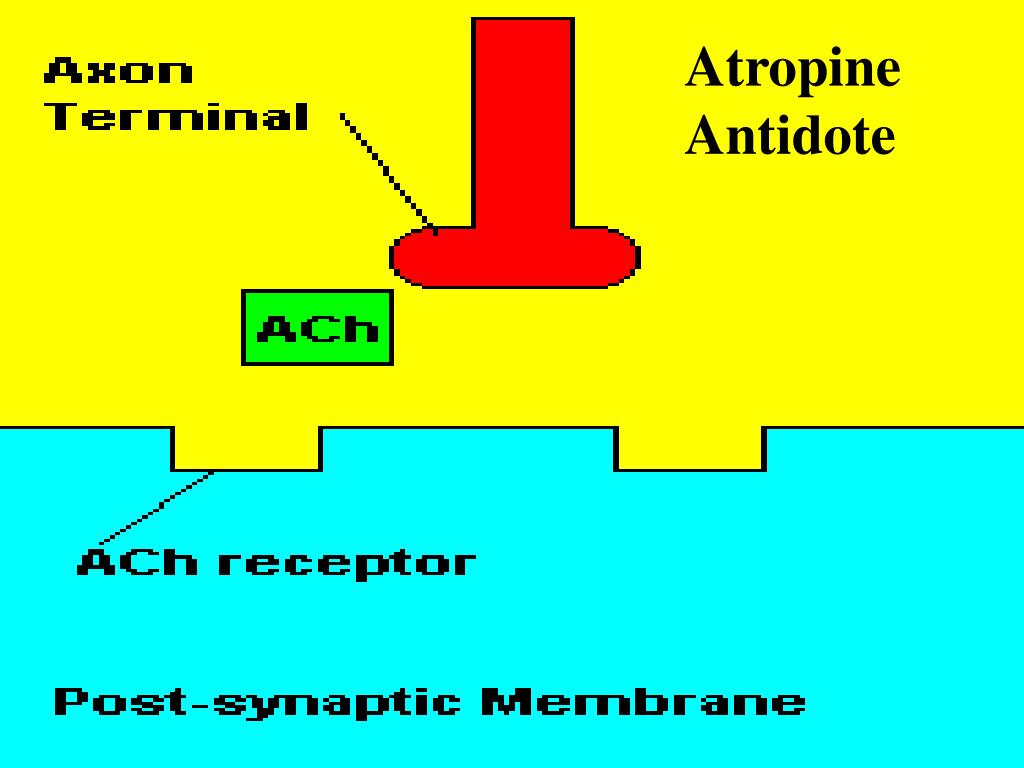
Following subcutaneous VX exposure, ATR/OBI-treated animals showed significant improvement in survival rate and progression of clinical signs compared to untreated animals. The PK of ATR and OBI administered either as a single drug, combined treatment but separately injected, or administered as the ATR/OBI co-formulation, was determined in plasma of naïve guinea pigs and found to be similar for all formulations. The present study evaluated the efficacy and pharmacokinetics (PK) of ATR/OBI following exposure to two different VX dose levels. Inhibition of AChE leads to overstimulation of the central and peripheral nervous system with convulsive seizures, respiratory distress and death as result. Organophosphates (OPs) irreversibly inhibit AChE, the enzyme responsible for termination of acetylcholine signal transduction. Nerve agent exposure is generally treated by an antidote formulation composed of a muscarinic antagonist, atropine sulfate (ATR), and a reactivator of acetylcholinesterase (AChE) such as pralidoxime, obidoxime (OBI), methoxime, trimedoxime or HI-6 and an anticonvulsant.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
